
CHOOSE AMYVID TO AID IN THE DIAGNOSIS OF ALZHEIMER’S DISEASE (AD)1,2
Amyvid provides objective evidence
Amyvid: PET imaging of the brain that estimates beta-amyloid plaque density in adult patients with cognitive impairment may aid in the diagnosis of AD.1-3
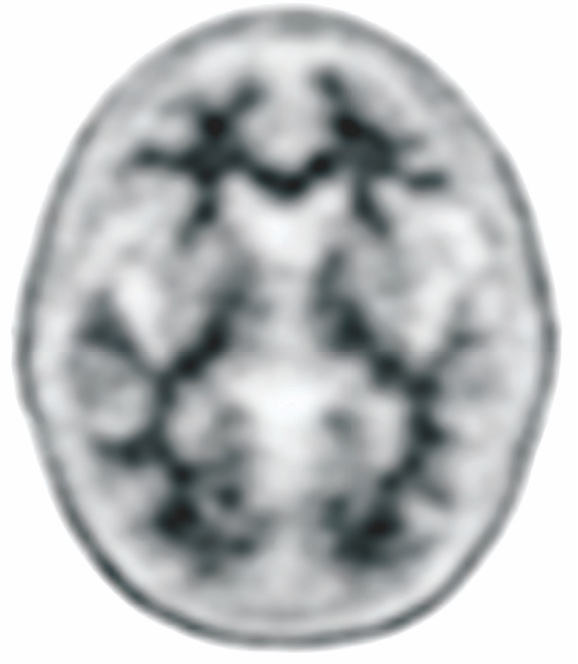
NEGATIVE AMYVID PET SCAN
POSITIVE AMYVID PET SCAN
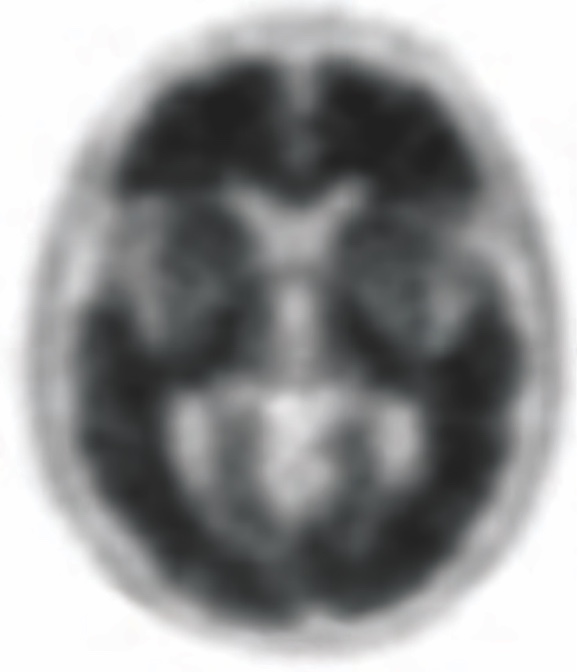
Amyvid scans1:
- Are interpreted using a binary visual read methodology (negative/positive)
- Are used as an adjunct to other diagnostic evaluations
- Should only be read by physicians who complete a comprehensive Amyvid training program
The objective of Amyvid image interpretation is to provide an estimate of the brain beta-amyloid neuritic plaque density, not to make a clinical diagnosis. Image interpretation is performed independently of a patient’s clinical features and relies upon the recognition of unique image features.1
Amyvid clinical studies:
- Amyvid was evaluated in 3 clinical studies that examined images from healthy adult subjects as well as subjects with a range of cognitive disorders, including some terminally ill patients who had agreed to participate in a postmortem brain donation program1
- All were single-arm studies in which subjects underwent an Amyvid injection and scan and then had images interpreted by multiple independent readers who were masked to all clinical information1
- Image interpretations used co-registration with CT scans when PET scans were performed on dual PET/CT scanners1
SELECT IMPORTANT SAFETY INFORMATION
Risk for Image Misinterpretation and Other Errors
Errors may occur during Amyvid image interpretation. Image interpretation should be performed independently of the patient’s clinical information. Amyvid scan results are indicative of plaque content only at the time of image acquisition and a negative scan does not preclude the development of brain amyloid in the future.
Learn more about the accuracy, reliability and reproducibility of Amyvid
SEE AMYVID EFFICACYCT=computed tomography; PET=positron emission tomography.
References:
- Amyvid (florbetapir F 18 injection) Prescribing Information. Lilly USA, LLC.
- McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7(3):263-269.
- Clark CM, Schneider JA, Bedell BJ, et al; for the AV45-A07 Study Group. Use of Florbetapir-PET for imaging β-amyloid pathology. JAMA. 2011;305(3):275-283.